 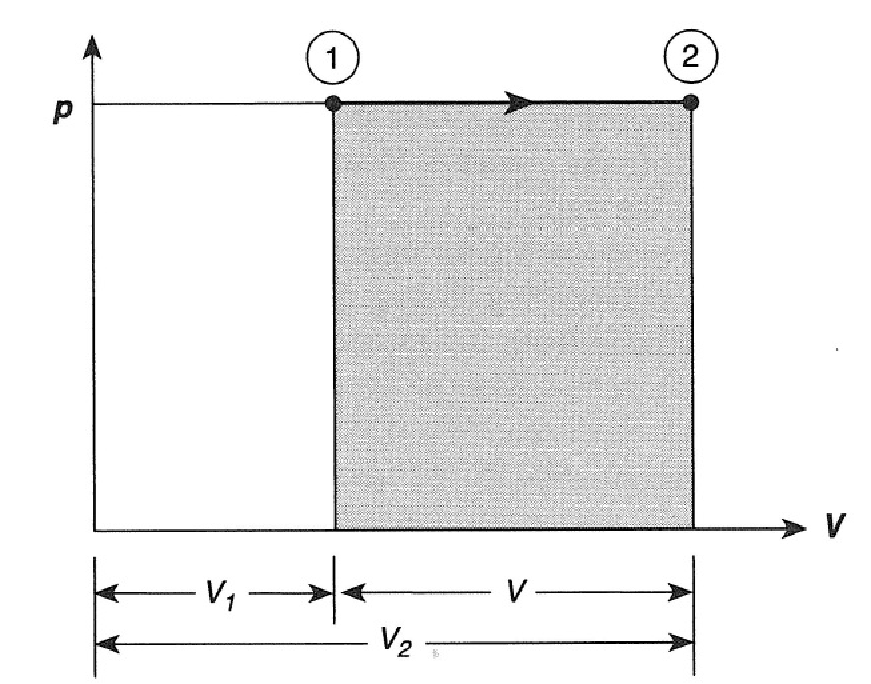
Different types of systems are generally characterized by different sets of variables. For an ideal gas, these variables are pressure, volume, temperature, and the number of molecules or moles of the gas. The thermal behavior of a system is described in terms of thermodynamic variables. Finally, with the interaction understood, we study the thermal behavior of the system with the help of the laws of thermodynamics. (We defined a system at the beginning of this chapter as anything whose properties are of interest to us it can be a single atom or the entire Earth.) Once our system is selected, we determine how the environment, or surroundings, interact with the system. We start by identifying the part of the universe we wish to study it is also known as our system. In thermodynamics, we take a similar approach. In solving mechanics problems, we isolate the body under consideration, analyze the external forces acting on it, and then use Newton’s laws to predict its behavior. Calculate physical quantities, such as the heat transferred, work done, and internal energy change for isothermal, adiabatic, and cyclical thermodynamic processes.Distinguish between quasi-static and non-quasi-static processes.By the end of this section, you will be able to: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
